All Myeloproliferative neoplasms (MPN) driver mutations converge on the activation of thrombopoietin receptor (MPL) signaling. Somatic frameshift mutations in calreticulin (CALR), leading to a novel peptide sequence are seen in 20-30% of patients with essential thrombocythemia or myelofibrosis. CALR is a multifunctional calcium-binding protein that is primarily localized to the endoplasmic reticulum (ER) of cells. It plays important roles in calcium homeostasis, protein folding, and quality control within the ER. MPN associated CALR mutants bind to and activate MPL resulting in constitutive activation of signaling.
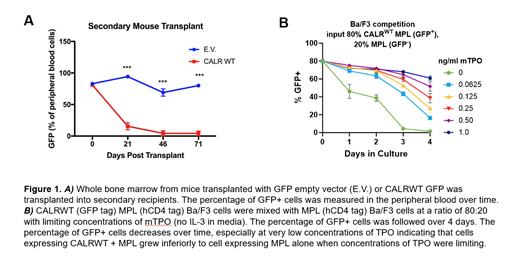
We established a transduction-transplantation model of MPN CALR mutant (52bp deletion, referred to as CALR DEL hereafter) MPN. As expected, mice transplanted with CALR DEL cells developed thrombocytosis, this phenotype was maintained in secondary transplants. We included mice transplanted with MSCV-IRES-GFP empty vector (EV) as well as CALRWT-IRES-GFP vector as controls. Abundant GFP + cells were seen in the peripheral blood and bone marrow of all control mice (EV and CALR WT) in primary transplants. In secondary transplants GFP + cells persisted in the EV cohort, however surprisingly no GFP + cells were seen in the CALR WT cohort. These experiments were repeated three independent times with similar results. We confirmed that the inability to serially transplant was not due to a homing defect. In addition, CALR WT cells demonstrated defective serial replating in methylcellulose.
The inability to serially transplant indicated that overexpression of CALR WT negatively impacts hematopoietic stem cell (HSC) self-renewal. To investigate how loss of CALR impacts hematopoiesis we utilized fetal liver (FL) cells from CALR -/- mice, the use of FL cells was required because CALR -/- is embryonic lethal. We performed competitive transplants with a 50:50 ratio of CD45.1/2 CALR -/- FL and CD45.1 CALR +/+ FL cells into CD45.2 donors. CALR -/-outcompeted CALR +/+ cells, demonstrating that loss of CALR enhances hematopoietic competitive potential.
We hypothesized that WT CALR negatively modulates MPL signaling, as the defective serially transplantation is also seen with MPL -/- and MPN associated CALR mutants are known to bind MPL and enhance signaling. To interrogate interactions between WT CALR and MPL we created Ba/F3 cells co-expressing mMPL (MSCV-IRES-hCD4 vector) and CALR WT, CALR DEL, or EV (MSCV-IRES-GFP vector). Cells co-expressing CALR WT and MPL cells had inferior growth compared to cells expressing MPL alone in limiting concentrations of thrombopoietin (TPO) (Figure 1B). The negative impact of CALR WT was unique to MPL dependent growth, as CALR WT + MPL cells grew equivalently well as MPL cells in limiting concentrations of IL-3.
In conclusion, we propose that CALR may serve a physiological function to modulate MPL signaling. Further studies are ongoing to interrogate direct interactions between CALR WT and MPL. Moreover, these features of CALR as a negative regulator of MPL signaling could be leveraged as a novel therapeutic approach in MPN to reduce oncogenic MPL signaling in MPN.
Disclosures
Fleischman:Pharmaessentia, CTI: Speakers Bureau; GSK, Incyte, CTI: Consultancy.